Entropy formula12/2/2023  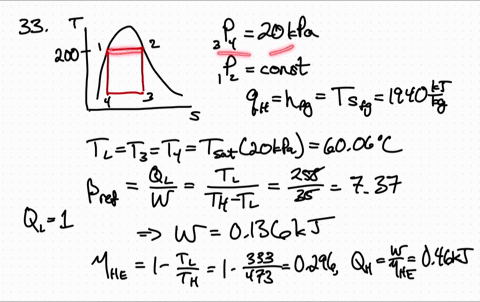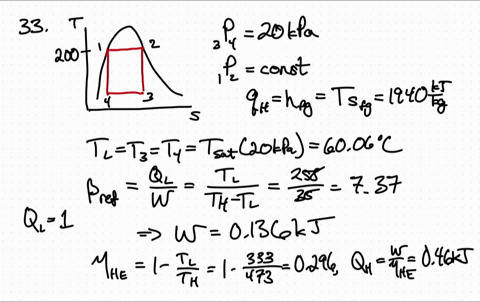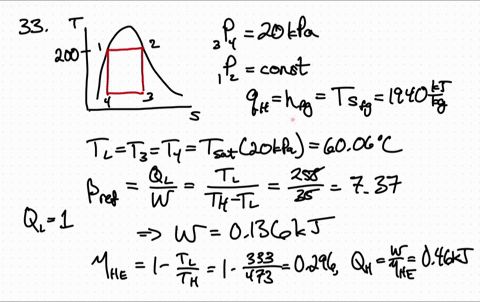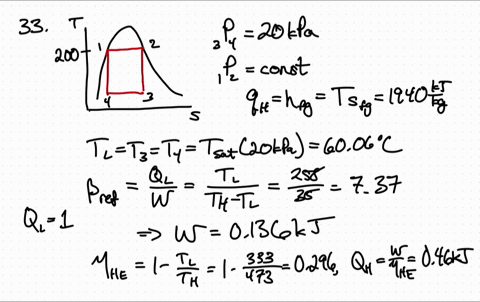
However, if the reaction occurs at high temperature the reaction becomes nonspontaneous, for the free energy change becomes positive when the high temperature is multiplied with a negative entropy as the enthalpy is not as large as the product. Entropy is zero in a reversible process it increases in an irreversible process. Lets use Shannon entropy formula in an example: You have a sequence of numbers: 1 0 3 5 8 3 0 7 0 1. If its 10, the unit is a dit, ban or hartley. When the base equals Eulers number, e, entropy is measured in nats. Another form of the second law of thermodynamics states that the total entropy of a system either increases or remains constant it never decreases. Our Shannon entropy calculator uses this base. 
When the reaction occurs at a low temperature the free energy change is also negative, which means the reaction is spontaneous. Entropy is the loss of energy available to do work. The temperature would also determine the spontaneous nature of a reaction if both enthalpy and entropy were positive. 
Because both enthalpy and entropy are negative, the spontaneous nature varies with the temperature of the reaction. Entropy is a measure of molecular disorder or randomness of a system, and the second law states that entropy can be created but it cannot be destroyed. ) There are several things worth noting about this equation. Using the entropy of formation data and the enthalpy of formation data, one can determine that the entropy of the reaction is -42.1 J/K and the enthalpy is -41.2 kJ. of thermodynamics In classical thermodynamics, entropy (from Greek o (trop) 'transformation') is a property of a thermodynamic system that expresses the direction or outcome of spontaneous changes in the system. This is the quantity that he called entropy, and it is represented by H in the following formula: H p1 log s (1/ p1) + p2 log s (1/ p2) + + pk log s (1/ pk ). Now one must find if the entropy is greater than zero to answer the question. If the enthalpy is negative then the reaction is exothermic. One may have to calculate the enthalpy of the reaction, but in this case it is given. \): Matrix of Conditions Dictating Spontaneity Case 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
